TNF_Superfamily_Pathway
发布时间:2019-12-11 14:56 来源:SABiosciences
- 通路
- 概述
Review
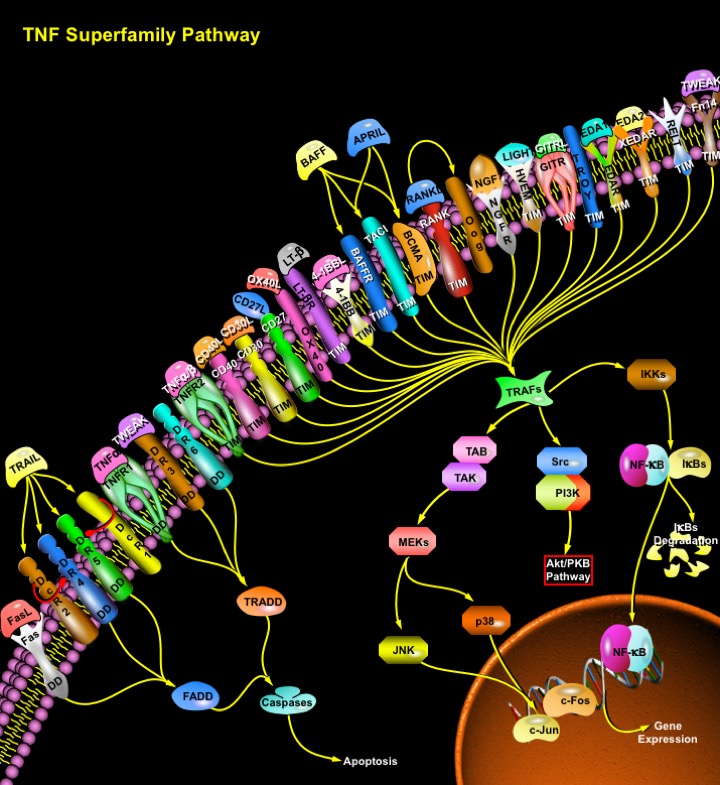
Members of the TNF (Tumor necrosis factor) receptor superfamily play pivotal roles in numerous biological events in metazoan organisms. Ligand-mediated trimerization by corresponding homo- or hetero-trimeric ligands, the TNF family proteins, causes recruitment of several intracellular adaptors, which activate multiple signal transduction pathways. 29 TNF receptor family members have been identified in humans. Based upon their cytoplasmic sequences and signaling properties, these TNF receptors can be classified into three major groups (Ref.1). The first group, including Fas/ CD95/ Apo1/ APT1, TNFR1/ CD120a /p55-R /TNFAR/ TNFR60, DR3/ TRAMP/ WSL1/ LARD /WSLLR/DDR3/TR3/Apo3, DR4/TRAILR1/Apo2, DR5/TRAILR2/KILLER/TRICK2A/TRICKB, and DR6/TR7, contains a DD (Death Domain) in the cytoplasmic tail. Fas, DR4 and DR5 interact with the FADD (Fas-Associated Death Domain) while TNFR1 and DR3 interact with the adaptor TRADD (TNFR-Associated Death Domain). These molecules, in turn cause activation of the Caspase cascade and induction of apoptosis (Ref.2).
The second group includes TNFR2/p75/CD120b/TNFR80/TNFBR, CD40/p50/Bp50, CD30/Ki-1/D1S166E, CD27/Tp55/S152, TNFR2-RP/TNFCR/TNFRIII, LT-BetaR, OX40/ CD134/ACT35/TXGP1L, 4-1BB/CD137/ILA, BAFFR, BCMA/BCM, TACI/CAML interactor, RANK/TRANCE-R, p75NGFR, HVEM (Herpes Virus Entry Mediator)/HveA/ATAR/TR2/ LIGHTR, GITR/AITR/ TNFRSF18, TROY/TAJ, EDAR, XEDAR/EDA-A2R, RELT and Fn14. These receptors contain one or more TIM (TRAF Interacting Motifs) in their cytoplasmic tails. Activation of TIM containing TNF receptors lead to recruitment of TRAF family members, and activation of multiple signal transduction pathways such as NF-KappaB (Nuclear Factor-KappaB), JNK (Jun N-terminal Kinase), p38, ERK (Extracellular Signal Regulated Kinase) and PI3K (Phosphoinisitide-3 Kinase) (Ref.3). The third group of TNF receptor family members, including DcR1/TRID/TRAIL-R3, DcR2/ TRUNDD/TRAIL-R4, DcR3 and Opg, does not contain functional intracellular signaling domains or motifs. Although this group of receptors cannot provide intracellular signaling, they can effectively compete with the other two signaling groups of receptors for their corresponding ligands. These DcR (Decoy Receptors) therefore function by impeding the activation of signal transduction pathways by other TNF receptors (Ref.4).
TRAFs are a major group of intracellular adaptors that bind directly or indirectly to many members of the TNF receptor superfamily. Six mammalian TRAFs, TRAF1 through TRAF6, have been identified. TRAFs can induce the activation of several kinase cascades that ultimately lead to the activation of signal transduction pathways such as NF-KappaB, JNK, ERK, p38 and PI3K, which can regulate cellular processes ranging from cell proliferation and differentiation to apoptosis. The TNFR superfamily contains a large number of proteins that regulate a very broad array of developmental and differentiating processes. Indeed, a number of biologic TNF blocking therapies are being used now to inhibit the inflammation associated with Crohn’s disease and rheumatoid arthritis. The continued examination of TNFR signal transduction will provide the tools for receptor or tissue specific interventions, allowing more targeted treatments that have fewer side effects.
References
- 1
- Dempsey PW, Doyle SE, He JQ, Cheng G. The signaling adaptors and pathways activated by TNF superfamily.
- 2
- Kischkel FC, Lawrence DA, Chuntharapai A, Schow P, Kim KJ, Ashkenazi A. Apo2L/TRAIL-dependent recruitment of endogenous FADD and caspase-8 to death receptors 4 and 5.
- 3
- Darnay BG, Ni J, Moore PA, Aggarwal BB. Activation of NF-kappaB by RANK requires tumor necrosis factor receptor-associated factor (TRAF) 6 and NF-kappaB-inducing kinase. Identification of a novel TRAF6 interaction motif.
- 4
- Gibson SB, Oyer R, Spalding AC, Anderson SM, Johnson GL. Increased expression of death receptors 4 and 5 synergizes the apoptosis response to combined treatment with etoposide and TRAIL.
 关于我们
关于我们