RNA_Polymerase_II_InitiationComplex
发布时间:2019-12-11 11:58 来源:SABiosciences
- 通路
- 概述
Review
The nuclei of all eukaryotic cells contain three different RNA Polymerases, designated I, II and III. Like the DNA Polymerase that catalyzes DNA replication, RNA Polymerases catalyze the formation of the phosphodiester bonds that link the nucleotides together to form a linear chain. The RNA Polymerase moves stepwise along the DNA, unwinding the DNA helix just ahead of the active site for polymerization to expose a new region of the template strand for complementary base-pairing. In this way, the growing RNA chain is extended by one nucleotide at a time in the 5’-to-3’ direction. The substrates are nucleoside triphosphates (ATP, CTP, UTP, and GTP); as for DNA replication, a hydrolysis of high-energy bonds provides the energy needed to drive the reaction forward. Each eukaryotic RNA Polymerase catalyzes transcription of genes encoding different classes of RNA. RNA Polymerase-II catalyzes transcription of all protein-coding genes; that is, it functions in production of mRNAs. RNA Polymerase-II also produces four snRNAs (small nuclear RNAs) that take part in RNA splicing.
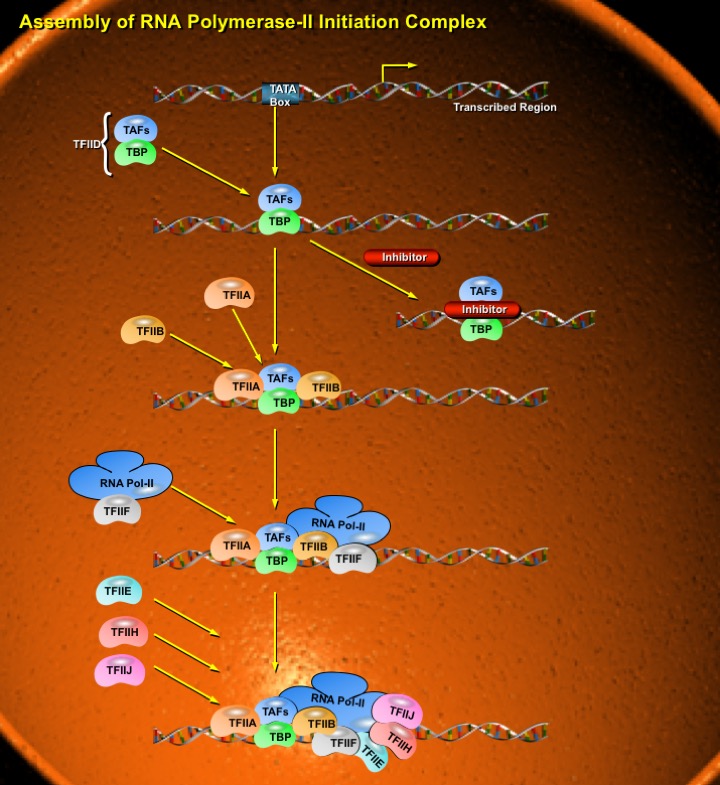
The eukaryotic polymerases do not directly recognize their core promoter sequences. The first step in complex formation at a promoter containing a TATA Box is binding of the factor TFIID to a region that extends upstream from the TATA se¬quence. TFIID is a complex made up of the TBP (TATA-Binding Protein), a small protein of -30 kD and at least 12 TAFs (TBP-Associated Factors). TBP is a sequence-specific protein and has a saddle-like shape that wraps partially around the double helix, forming a platform onto which the remainder of the initiation complex can be assembled (Ref.1). TBP binds DNA in an unusual way. It not only sits in the minor groove (virtually all known DNA-binding proteins bind in the wide groove), but also bends the DNA by -80°. The TATA Box bends to¬wards the major groove, widening the minor groove. The distortion is restricted to the 8bp of the TATA Box; at each end of the sequence, the minor groove has its usual width of ~5 A, but at the center of the sequence the minor groove is >9 A. This is a deformation of the structure, but does not actually separate the strands of DNA, because base pairing is maintained. This structure has several functional implications. By changing the spatial organization of DNA on either side of the TATA Box, it allows the transcription factors and RNA Polymerase to form a closer association than would be possible on linear DNA. The bending at the TATA box corresponds to unwinding of about 1/3 of a turn of DNA, and is compensated by a positive writhe. The presence of TBP in the minor groove, combined with other proteins binding in the major groove, cre¬ates a high density of protein-DNA contacts in this re¬gion. The TAFs are intriguing proteins that appear to play a variety of roles during initiation of transcription and also during other events that involve assembly of multiprotein complexes onto the genome. Some TAFs are stoichiometric with TBP; others are present in lesser amounts. TFIIDs containing dif¬ferent TAFs could recognize different promoters. Some (substoichiometric) TAFs are tissue-specific. The total mass of TFIID typically is -800 kD, containing TBP and 11 TAFs, varying in mass from 30-250 kD.
During transcription, TAFs assist in attachment of TFIID to the TATA Box and, in conjunction with other proteins called TICs (TAF-and Initiator-Dependent Cofactors); also participate in recognition of the Initiator sequence, especially at those promoters that lack a TATA Box. After TFIID has attached to the core promoter, the PIC (Pre-Initiation Complex) is formed by attachment of the remaining GTFs (General Transcription Factors) (Ref.2). These GTFs bind to the complex in the order TFIIA, TFIIB, TFIIF/ RNA Polymerase-II, TFIIE, TFIIH and TFIIJ. The assembly involves a complex set of interactions (Ref.3). Within the overall process, three events are particularly important:(1) Attachment of TBP induces formation of a bend in the DNA in the region of the TATA Box, (2) The bend provides a recognition structure for TFIIB, which ensures correct positioning of RNA Polymerase-II relative to the transcription start site, and (3) The disruption to the base pairing needed to form the open promoter complex is brought about by TFIIH (Ref.4). The final step in assembly of the Initiation Complex is the addition of phosphate groups to the CTD (C-Terminal Domain) of the largest subunit of RNA Polymerase-II. In mammals, this domain consists of 52 repeats of the seven-amino-acid sequence Tyr-Ser-Pro-Thr-Ser-Pro-Ser (Ref.5 & 6). Two of the three serines in each repeat unit can be modified by addition of a phosphate group, causing a substantial change in the ionic properties of the polymerase. Once phosphorylated, the polymerase is able to leave the PIC and begin synthesizing RNA. Phosphorylation might be carried out by TFIIH, which is one of the last factors to add to the complex. TFIIH has DNA Helicase activity and can use the energy from hydrolysis of ATP to separate the strands of the duplex template DNA. This protein is suspected to mediate unwinding of the strands at the start site, allowing the polymerase to initiate transcription. TFIIH also has a protein kinase activity, which can transfer the Gamma-phosphates of ATPs to multiple serine in the C-Terminal repeat domain of the largest RNA Polymerase-II subunit (Ref.3) After departure of the polymerase, at least some of the GTFs detach from the core promoter, but TFIID, TFIIA and TFIIH remain, enabling re-initiation to occur without the need to rebuild the entire assembly from the beginning (Ref.4). Re-initiation is therefore a more rapid process than primary initiation, which means that once a gene is switched on, transcripts can be initiated from its promoter with relative ease until such a time as a new set of signals, switches the gene off.
Assembly of the RNA Polymerase-II initiation com¬plex provides an interesting contrast with prokaryotic transcription. Bacterial RNA Polymerase is essentially a coherent aggregate with intrinsic ability to bind DNA; the Sigma Factor, needed for initiation but not for elongation, becomes part of the enzyme before DNA is bound, although it is later released. But RNA Polymerase-II can bind to the promoter only after separate transcription factors have bound. The factors play a role analogous to that of bacterial Sigma Factor-to allow the basic Polymerase to recognize DNA specifically at promoter sequences-but have evolved more independence. Indeed, the factors are primarily responsible for the specificity of promoter recognition.
References
- 1
- Chasman DI, Flaherty KM, Sharp PA, Kornberg RD. Crystal structure of yeast TATA-binding protein and model for interaction with DNA.
- 2
- Szutorisz H, Dillon N, Tora L. The role of enhancers as centres for general transcription factor recruitment.
- 3
- Lee TI, Young RA. Transcription of eukaryotic protein-coding genes.
- 4
- Kim TK, Ebright RH, Reinberg D. Mechanism of ATP-dependent promoter melting by transcription factor IIH.
- 5
- Lux C, Albiez H, Chapman RD, Heidinger M, Meininghaus M, Brack-Werner R, Lang A, Ziegler M, Cremer T, Eick D. Transition from initiation to promoter proximal pausing requires the CTD of RNA polymerase II.
- 6
- Chapman RD, Palancade B, Lang A, Bensaude O, Eick D. The last CTD repeat of the mammalian RNA polymerase II large subunit is important for its stability.
 关于我们
关于我们