RhoA_Pathway
发布时间:2019-12-11 11:58 来源:SABiosciences
- 通路
- 概述
Review
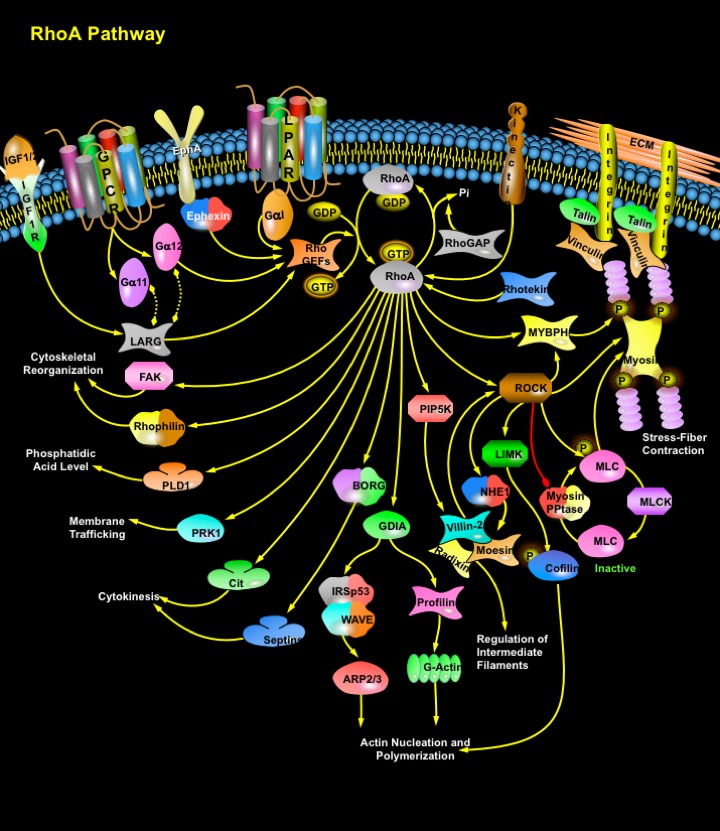
Rho is a member of the Ras superfamily of small GTP-binding proteins that play a central role in diverse biological processes such as Actin cytoskeleton organization, Microtubule dynamics, Gene transcription, Oncogenic transformation, Cell cycle progression, Adhesion and Epithelial wound repair. To date, 20 genes encoding different members of the Rho family have been identified in the human genome, and each one acts as a molecular switch to control distinct biochemical pathways. The mammalian Rho GTPase family currently consists of three subfamilies, Rho (RhoA, RhoB and RhoC), Rac (Rac1, Rac2 and Rac3) and CDC42 (Cell Division Cycle-42) (CDC42Hs and G25K). Rho proteins cycle between an active GTP-bound state and an inactive GDP-bound state. Their activation state is controlled by regulatory proteins such as GEFs (Guanine Exchange Factors), which catalyze the exchange of GDP for GTP thereby activating Rho, GDIs (Guanine Dissociation Inhibitors), which inhibit the release of GDP keeping Rho inactive, and GAPs (GTPase Activating Proteins), which increase the rate at which Rho hydrolyzes GTP and hence becomes inactivated. While the overall sequence of events leading to activation of Rho family proteins by extracellular signals is known, gaps remain in the molecular details of these pathways and are areas of intensive study (Ref.1, 2 & 3).
Higher vertebrates have 3 Rho GTPases: RhoA, RhoB, and RhoC, which share 85% amino acid sequence identity. Although RhoB and RhoC were characterized at the same time as RhoA they have received less attention because of their extensive homology to RhoA. The RhoA gene is longer and contains more exons and introns than RhoC. The RhoB gene is far smaller and contains only one exon, and is thought to have arisen by reverse transcription. RhoA, the most extensively studied Rho Family member, is activated by a variety of Growth factors, Cytokines, Adhesion molecules, Hormones, Integrins, G-proteins and other biologically active substances. The major activator of RhoA is GPCR (G-Protein coupled Receptor). GPCRs that activate RhoA and use G-Alpha11, G-Alpha12 or G-AlphaI for signal transduction include receptors for LPA (Lysophosphatidic Acid) and certain Hormones. EphA (EphrinA) Receptors can also directly activate RhoA through Ephexin. IGF (Insulin-Like Growth Factor) can also activate RhoA indirectly. Upon binding with IGF1/2, IGF1R (Insulin-Like Growth Factor-I Receptor) forms a complex with LARG (Leukemia-Associated RhoGEF-12), which in turn activates RhoA. RhoA is also activated by Ktn1 (Kinectin-1). Upon activation, RhoA interacts with and regulate a spectrum of functionally diverse downstream effectors, initiating a network of cytoplasmic and nuclear signaling cascades (Ref.1, 4 & 5).
A number of proteins have been identified as targets of RhoA. These targets include the PAK (p21-Activated Kinase) family, Rho-kinase/ROK/ROCK (Rho-Associated Coiled-Coil-Containing Protein Kinase), MBS (Myosin-Binding Subunit) of Myosin PPtase (Myosin Phosphatase), PKN (Protein Kinase-N)/PRK-1, Rhophilin, Rhotekin, Citron, and GDIA. ROCK is the major target of RhoA. RhoA is important for the organization of stress fibers and also in the regulation of acto-myosin contractility through Myosin PPtase (Myosin Phosphatase), MLCP (Myosin Light Chain Phosphatase) phosphorylation and through ROCK. Integrins stimulate the production of PIP2 (Phosphatidyl Inositol Biphosphate) and this effect is mediated by RhoA through its interaction with a Type I isoform of PIP4, 5K (Phosphatidyl Inositol-4-Phosphate-5-Kinase). The increase in PIP2 synthesis by RhoA is potentially relevant to focal adhesion assembly because the actin binding activity of several cytoskeletal proteins such as Profilin, Gelsolin and Vinculin is modulated by PIP2 enriched in focal adhesion plaques. In addition, Rho-GTP also regulates Integrin-clustering by disrupting the Integrin clustering and focal plaque formation. ROCK also activates LIMK (LIM-Kinase). Both LIMK1 and 2 phosphorylate and inactivate Cfl (Cofilin), an Actin-depolymerizing factor, and regulates Actin Cytoskeletal reorganization. ROCK, can also phosphorylate ERM (Ezrin/ Vil (Villin), Rdx (Radixin) and Msn (Moesin)) proteins in vitro but the effect of ROCK inhibitors on ERM phosphorylation in vivo varies and it is unclear whether ROCK acts directly or indirectly to modify ERM phosphorylation. ROCK can also phosphorylate the sodium–hydrogen exchanger, NHE1 (Na-H Exchanger-1). NHE1, on the other hand, interacts with ERM proteins both directly and via EBP50 (Ezrin-Radixin-Moesin Binding Phosphoprotein-50). ERM Proteins can also be activated by Rho via PIP5K (Phosphatidylinositol-4-Phosphate 5-Kinase). Both Rac and Rho bind to and activate PIP5K, which increases the amount of PIP2 (Phosphatidylinositol (4,5)-bisphosphate). PIP2 then activates ERM proteins by inhibiting their interdomain interaction, which allows phosphorylation of their C-terminal threonine residue by some kinases. The C-terminally threonine-phosphorylated ERM proteins are stabilized at the activated forms, which function as Actin filament/plasma membrane cross-linkers to form microvilli. Activated ERM proteins are associated directly with the Adhesion molecules such as CD44 and ICAM-1 (Intercellular Adhesion Molecule-1), -2, and -3 and indirectly with other integral membrane proteins such as NHE3 (Sodium/Hydrogen Exchanger-3) through EBP50/NHERF (Sodium-Hydrogen Exchanger Regulatory Factor). Activated ERM proteins also bind to Rho-GDI at their N-terminal halves, suppressing GDI activity of Rho-GDI to release GDP-Rho, which is activated to GTP-Rho. This GTP-Rho can be used to activate ERM proteins just beneath the plasma membranes, providing a positive feedback pathway. Other reported substrates for Rho kinases include MARCKS, EF-1Alpha, Calponin, and CPI-17 and Collapsin-Response Mediator Protein-2 (Ref. 6, 7 & 8).
Besides ROCK, other important targets of Rho include FAK (Focal Adhesion Kinase), PRK-1/ PKN1, BORG (Binder of Rho GTPases), Citron, PLD (Phospholipase-D) and GDIA. The GTPase RhoA plays a prominent role in regulating the organization of the cytoskeleton by promoting the assembly of focal adhesions and actin stress fibers and by activating FAK. PKN1/PRK-1 and PKN2 are Rho effectors involved in endosomal trafficking. Citron is a ROCK-related kinase that is critical for cytokinesis and is also implicated in other aspects of cell cycle progression. BORG proteins are Rho effectors that connect to Septs (Septins), structural proteins that can polymerize to form filaments involved in cytokinesis in yeast and mammalian cells, and that probably carry out additional structural roles in mammalian cells as well. Members of the Rho subfamily of GTP-binding proteins including Rho and Rac are implicated in the regulation of PLD. PLD catalyzes the hydrolysis of Phosphatidylcholine to yield Phosphatidic Acid and Choline. Phosphatidic Acid is a second messenger involved in membrane remodeling events that are critical to cell growth, such as vesicle trafficking and regulated secretion. Rho also activates scaffolding proteins such as GDIA, WASP (Wiskott-Aldrich syndrome protein) and IRSp53 (Insulin Receptor Substrate Protein-53). RhoA also binds to Rhophilin and regulate the actin cytoskeleton. RhoA also interacts with a Rho effector protein, Rhotekin. The RBD (Rho Binding Domain) motif of Rhotekin binds specifically to the GTP-bound form of RhoA. The fact that the RBD region of Rhotekin has a high affinity for GTP-RhoA and that Rhotekin binding results in a significantly reduced intrinsic and catalytic rate of GTP hydrolysis make it an ideal tool for affinity purification of GTP-RhoA from cell lysates (Ref. 2, 9 & 10). RhoA dependent signaling pathway is recognized as an essential regulator of vascular functions, and seems to play an important role in major arterial diseases such as hypertension, atherosclerosis and pulmonary hypertension. More studies are now needed to understand how RhoA is activated, what its downstream effectors are and how this complex signaling pathway regulates smooth muscle cell functions in physiological conditions as well as in association with vascular diseases. However, the present knowledge of Rho signaling suggests that pharmacological agents targeting the RhoA/Rho kinase signaling pathway may have therapeutic benefits in arterial diseases (Ref.9, 10 & 11).
References
- 1
- Loirand G, Guilluy C, Pacaud P. Regulation of Rho proteins by phosphorylation in the cardiovascular system.
- 2
- Fessler MB, Arndt PG, Just I, Nick JA, Malcolm KC, Worthen GS. A dual role for RhoA in suppression and induction of cytokines in the human neutrophil.
- 3
- Boureux A, Vignal E, Faure S, Fort P. Evolution of the Rho Family of Ras-like GTPases in Eukaryotes.
- 4
- Stevenson AS, Matthew JD, Eto M, Luo S, Somlyo AP, Somlyo AV. Uncoupling of GPCR and RhoA-induced Ca2+-sensitization of chicken amnion smooth muscle lacking CPI-17.
- 5
- Santama N, Er CP, Ong LL, Yu H. Distribution and functions of kinectin isoforms.
- 6
- Miyazaki K, Komatsu S, Ikebe M. Dynamics of RhoA and ROKalpha translocation in single living cells.
- 7
- Nakamura Y, Kaneto H, Miyatsuka T, Matsuoka TA, Matsuhisa M, Node K, Hori M, Yamasaki Y. Marked increase of insulin gene transcription by suppression of the Rho/Rho-kinase pathway.
- 8
- Sato J, Iiri T. Rho and Rho-kinase
- 9
- Ito H, Iwamoto I, Mizutani K, Morishita R, Deguchi T, Nozawa Y, Asano T, Nagata K. Possible interaction of a Rho effector, Rhotekin, with a PDZ-protein, PIST, at synapses of hippocampal neurons.
- 10
- Peck JW, Oberst M, Bouker KB, Bowden E, Burbelo PD. The RhoA-binding protein, rhophilin-2, regulates actin cytoskeleton organization.
 关于我们
关于我们