CDK5_Pathway
发布时间:2019-12-09 19:50 来源:SABiosciences
- 通路
- 概述
Review
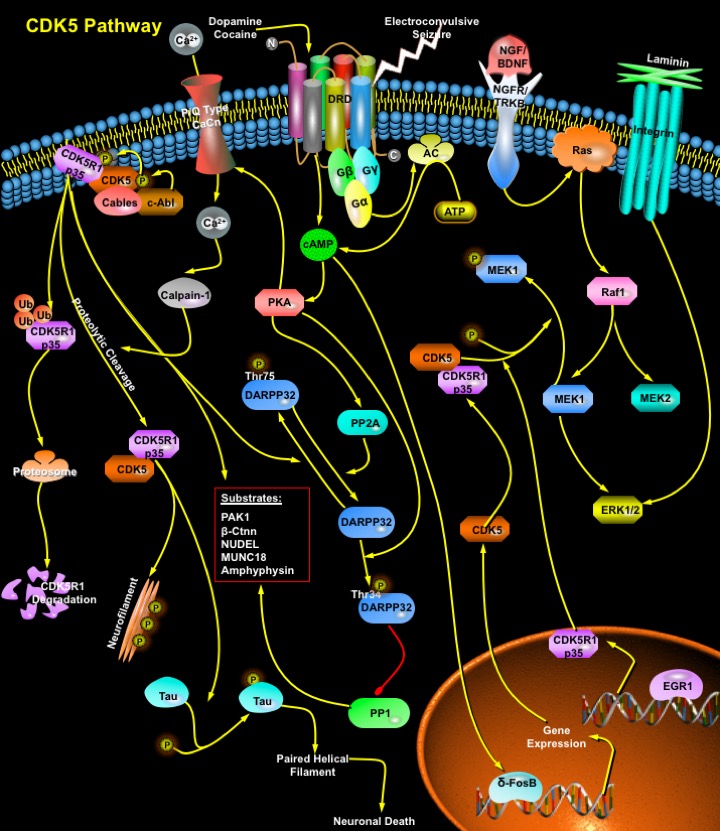
CDKs (Cyclin-dependent kinases) are a group of serine/threonine protein kinases activated by binding to a regulatory subunit cyclin. These kinases are key regulators of the eukaryotic cell cycle progression. CDK5 (also known as Neuronal CDC2-Like Kinase, NCLK) is a unique member of the CDK family, that does not act as a checkpoint kinase to regulate cell cycle progression, but acts as a regulatory kinase involved in other post-mitotic processes such as neuronal activity, neuronal migration during development and neurite outgrowth. Although the CDK5 protein is expressed at basal levels in most mammalian tissues, CDK5 activity is also seen in brain and developing muscle, as these are the only tissues that express CDK5 activators. Activation of CDK5 requires its association with a regulatory subunit, and CDK5 activator, p35/CDK5R1 (NCK5a, Neuronal CDK5 activator) (Ref.1). A deficiency of CDK5 or CDK5R1 results in a reversed layer formation of cortical neurons in the cerebrum or a defect in the formation of the cortical laminar structure in the cerebellum, p35-CDK5 is thought to play an important role in brain development (Ref.2). Over 20 functionally diverse proteins involved in cytoskeleton dynamics, cell adhesion, transport, and membrane trafficking act as CDK5 substrates elucidating the molecular mechanisms of CDK5 function (Ref.1).
CDK5 is recruited to the membrane through its interaction with the activator p35, which is myristoylated. p35 is a short-lived protein, and its phosphorylation by CDK5 targets it for ubiquitin-mediated proteolysis. Calpain1 also directly cleaves p35 to release a fragment of 25KD. Post-translational modifications, such as phosphorylation by c-Abl (through an Abl-binding adaptor protein called Cables), regulate CDK5 kinase activity. Engagement of the integrin receptors by Laminin stimulates transcription of p35. NGF (Nerve Growth Factor) and BDNF (Brain-Derived Neurotrophic Factor) bind to receptors (NGFR and TRKB) and induce the ERK (Extracellular Signal-Regulated Kinase)-mediated expression of p35, which requires the transcription factor EGR1 (Early Growth Response 1) (Ref.3). Proteolytic cleavage of p35 produces p25, which accumulate in the brains of patients with Alzheimer's disease. Conversion of p35 to p25 causes prolonged activation and mislocalization of CDK5 and hyperphosphorylation of Tau (a neuron-specific MAP), leading to the formation of paired helical filaments and promotes apoptosis. One target of CDK5 is MEK1 (MAPK/ERK kinase-1). Phosphorylation of MEK1 by CDK5 represses MEK1 activity and blocks downstream cellular responses. The activation of p35 by MAPK (Mitogen-Activated Protein Kinase) pathways followed by deactivation of MAPK signaling by the CDK5/p35 complex completes the loop of a feedback circuit to terminate MAPK signaling. CDK5 induces PAK1 (p21-Activated Kinase) hyperphosphorylation in a Rac-dependent manner, which results in downregulation of PAK1 kinase activity. CDK5 is also associated with the microtubule cytoskeleton. Phosphorylation of Tau by CDK5 reduces the binding of Tau to microtubules, inhibits the ability of Tau to promote microtubule assembly and decreases microtubule-nucleation activity. Modulation of microtubule stability by phosphorylation of several MAPs (Microtubule Associated Proteins) is part of the programme by which CDK5 exerts its effect on neuronal migration (Ref.3).
The p35–CDK5 complex associates with Beta-Ctnn (Beta-catenin) and N-Cadherin, and inhibition of CDK5 activity cause an increase in N-Cadherin-mediated aggregation of neurons. Conversely, active CDK5 dissociates Beta-Ctnn from N-Cadherin, which is accompanied by a loss of adhesion. Beta-Ctnn is a substrate of CDK5, and phosphorylation by CDK5 regulates its association with the Cadherins (Ref.3). CDK5 and p35 also are enriched at the neuromuscular junction, where CDK5 activity is required for the Neuregulin-induced transcription of Acetylcholine receptors. CDK5-mediated phosphorylation of Synapsin-1, MUNC18, Amphiphysin-1, and the Alpha-subunit (1A) of P/Q-type VDCC (Voltage-Dependent Ca2+ Channels) plays a role in synaptic vesicle trafficking and neurotransmitter release. CDK5 is also involved in postsynaptic transmission, and modulates dopaminergic signaling through DRD (Dopamine Receptor) in the neostriatal region of the brain. DRD is a G-protein Coupled Receptor stimulates AC (Adenylyl Cyclase) and activates cAMP-dependent protein kinases,which regulate neuronal growth and development, mediate some behavioral responses, and modulate DRD-mediated events. The transcription factor DeltaFosB promotes CDK5 expression in response to chronic Cocaine and ECS (Electroconvulsive Seizure) treatments, implicating the dopaminergic signaling pathway. The role of CDK5 in Dopamine responses occurs through phosphorylation of DARPP32 (Dopamine- and cAMP-Regulated Phosphoprotein), a neostriatum-specific protein. Depending on its phosphorylation state, DARPP32 inhibits either PP1 (Protein Phosphatase-1) or PKA (Protein Kinase-A). Phosphorylation of DARPP32 by PKA on Thr34 makes it a potent inhibitor of PP1, and potentiates Dopamine-induced phosphorylation of PKA substrates. In contrast, phosphorylation of DARPP32 at Thr75 by CDK5 renders it an inhibitor of PKA. PP1 is also phosphorylated by both PKA and CDK5 with antagonistic consequences. Phosphorylation of DARPP32 by PKA converts it into an inhibitor of PP1, whereas phosphorylation by CDK5 makes it a worse substrate for PKA. So, CDK5 and PKA exert opposing effects on Dopamine signaling through phosphorylation of DARPP32, PP1 and possibly other proteins in the pathway (Ref.4 & 7). CDK5 activity has also been implicated in the mGluR-mediated enhancement of voltage-dependent Ca2+ currents.
Since it was identified a decade ago, CDK5 has emerged as a crucial regulator of neuronal migration in the developing CNS (Central Nervous System). CDK5 phosphorylates a diverse list of substrates, implicating it in the regulation of a range of cellular processes. The best known roles for CDK5 is in the regulation of cytoarchitecture of the CNS, regulation of the cytoskeleton, axon guidance, membrane transport, synaptic function, Dopamine signaling and drug addiction. Deregulation of CDK5 has been linked to the pathology of neurodegenerative diseases, such as Alzheimer's disease and ALS (Amyotrophic Lateral Sclerosis) (Ref.3). CDK5 also has been implicated in the pathological degeneration of neurons. Dysregulation of CDK5 causes the hyperphosphorylation of Tau, thereby contributing to neurofibrillary tangle formation (Ref.5 & 8). p35–CDK5 is expressed in lens epithelial cells and in differentiating lens fibers. CDK5 is expressed in Leydig cells, Sertoli cells, spermatogonia and peritubular cells indicating a role in spermatogenesis. CDK5 is also expressed in insulin-secreting pancreatic cells, and inhibition of CDK5 reduces insulin secretion, indicating that CDK5 might be a positive regulator of insulin exocytosis (Ref.3). Moreover, it has recently been reported that CDK5 phosphorylated NUDEL, a novel LIS1-interacting protein in the growth cone. The inhibition of CDK5 activity alters NUDEL localization and its association with the dynein complex, resulting in the alteration of neuronal migration (Ref.6).
References
- 1
- Wang CX, Song JH, Song DK, Yong VW, Shuaib A, Hao C. Cyclin-dependent kinase-5 prevents neuronal apoptosis through ERK-mediated upregulation of Bcl-2.
- 2
- O
- 3
- Dhavan R, Tsai LH. A decade of CDK5.
- 4
- Wei FY, Nagashima K, Ohshima T, Saheki Y, Lu YF, Matsushita M, Yamada Y, Mikoshiba K, Seino Y, Matsui H, Tomizawa K. Cdk5-dependent regulation of glucose-stimulated insulin secretion.
- 5
- Saito T, Onuki R, Fujita Y, Kusakawa G, Ishiguro K, Bibb JA, Kishimoto T, Hisanaga S. Developmental regulation of the proteolysis of the p35 cyclin-dependent kinase 5 activator by phosphorylation.
- 6
- . Tomizawa K, Ohta J, Matsushita M, Moriwaki A, Li ST, Takei K, Matsui H. Cdk5/p35 regulates neurotransmitter release through phosphorylation and downregulation of P/Q-type voltage-dependent calcium channel activity.
- 7
- Hamada M, Hendrick JP, Ryan GR, Kuroiwa M, Higashi H, Tanaka M, Nairn AC, Greengard P, Nishi A. Nicotine Regulates DARPP-32 (Dopamine- and cAMP-Regulated Phosphoprotein of 32 kDa) Phosphorylation at Multiple Sites in Neostriatal Neurons.
- 8
- Ahn JS, Radhakrishnan ML, Mapelli M, Choi S, Tidor B, Cuny GD, Musacchio A, Yeh LA, Kosik KS. Defining Cdk5 ligand chemical space with small molecule inhibitors of tau phosphorylation.
 关于我们
关于我们