Chromatin_Remodeling
发布时间:2019-12-10 09:39 来源:SABiosciences
- 通路
- 概述
Review
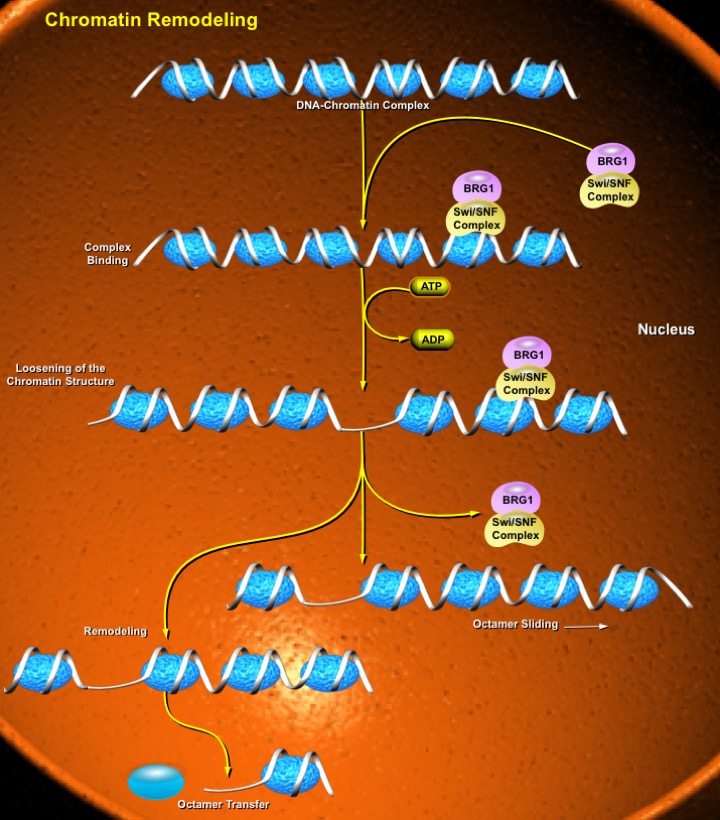
The condensation of DNA into an ordered chromatin structure allows the cell to solve the topological problems associated with storing huge molecules of chromosomal DNA within the nucleus. DNA is packaged into chromatin in orderly repeating protein-DNA complexes called nucleosomes. Each nucleosome consists of approximately 146bp of dsDNA (double-stranded DNA) wound 1.8 times around a histone octamer (Ref.1). Two molecules each of H2A, H2B, H3, and H4 comprise the histone ramp around which the DNA superhelix winds. Stretches of DNA upto 100bp separate adjacent nucleosomes. Multiple nuclear proteins bind to this linker region, some of which may be responsible for the ordered wrapping of strings of nucleosomes into higher-order chromatin structures (Ref.2).
Chromatin assembly involves the formation of nucleosomes from histones and DNA, whereas chromatin remodeling involves the disruption and re-formation of histone–DNA contacts. Chromatin remodeling factors comprise an ATPase subunit along with other polypeptides that are responsible for the regulation, efficiency, and functional specificity of each complex. The ATPase subunit belongs to the Snf2 (Sucrose Nonfermenting in Yeast) super family of proteins, which includes the SWI2/Snf2 group and the imitation SWI (ISWI) group. The SWI2/Snf2 group includes yeast SWI/Snf (ySWI/Snf), yeast RSC, the Drosophila Brm (Brahma) complex, and the hBrm (human Brm) and BRG1 (Brm/SWI2 Related Gene-1, hBRG1 in humans) complexes (Ref.3). Moreover, there are many other proteins that are closely related to the ATPase subunits of chromatin remodeling complexes. In order to remodel chromatin, remodeling complexes recognize and bind to their substrate. The SWI/Snf, NURF, and RSC complexes act as ATP-dependent motors that track along the DNA strands and pull them away from the histone octamer cores. During this shift of histone-DNA contact points, the DNA presumably becomes accessible to the transcriptional machinery. These complexes serve to maintain chromatin in a repressive configuration by dissociating other chromatin-associated proteins from the DNA, such as the errant TATA-binding protein (Ref.2). Another class of chromatin-altering factor acts by covalently modifying histone proteins. These modifications include phosphorylation, ubiquitination, ADP-ribosylation, and methylation, but the best-characterized mechanism is acetylation, catalyzed by Histone Acetyl-Transferase enzymes (Ref.1). The exact consequence of remodeling is dependent on the exact context of nucleosomes at a given promoter and can lead to either (i) activation of transcription or (ii) repression (Ref.3). In addition to catalyzing nucleosome mobility, chromatin-remodeling factors can enhance the access of DNA-binding factors and nucleases to DNA packaged into chromatin. These activities are also consistent with the ability of remodeling factors to disrupt histone–DNA interactions in the nucleosome (Ref.2). Disruption of these mechanisms gives rise to transcriptional chaos and leukemic transformation such as APL (French-American-British [FAB] M3).
Syndromes of disordered 'chromatin remodeling' are unique in medicine because they arise from a general deregulation of DNA transcription caused by mutations in genes encoding enzymes which mediate changes in chromatin structure. These mutations result in a loss of proper regulation of chromatin structure, which in turn causes deregulation of gene transcription and inappropriate protein expression. Some of the genetic diseases that arise as a consequence of disordered chromatin remodeling include: Alpha-Thalassemia/Mental Retardation Syndrome, ATR-X (ATM/Rad3-related); Rett Syndrome (RS); ICF (Immunodeficiency-Centromeric Instability-Facial Anomalies Syndrome); RSTS (Rubinstein-Taybi Syndrome), and CLS (Coffin-Lowry Syndrome) (Ref.4).
References
- 1
- Sterner DE, Berger SL. Acetylation of histones and transcription-related factors.
- 2
- Redner RL, Wang J, Liu JM. Chromatin remodeling and leukemia: new therapeutic paradigms.
- 3
- Vignali M, Hassan AH, Neely KE, Workman JL. ATP-dependent chromatin-remodeling complexes.
- 4
- Ausio J, Levin D, De Amorim G, Bakker S, Macleod P. Syndromes of disordered chromatin remodeling.
 关于我们
关于我们